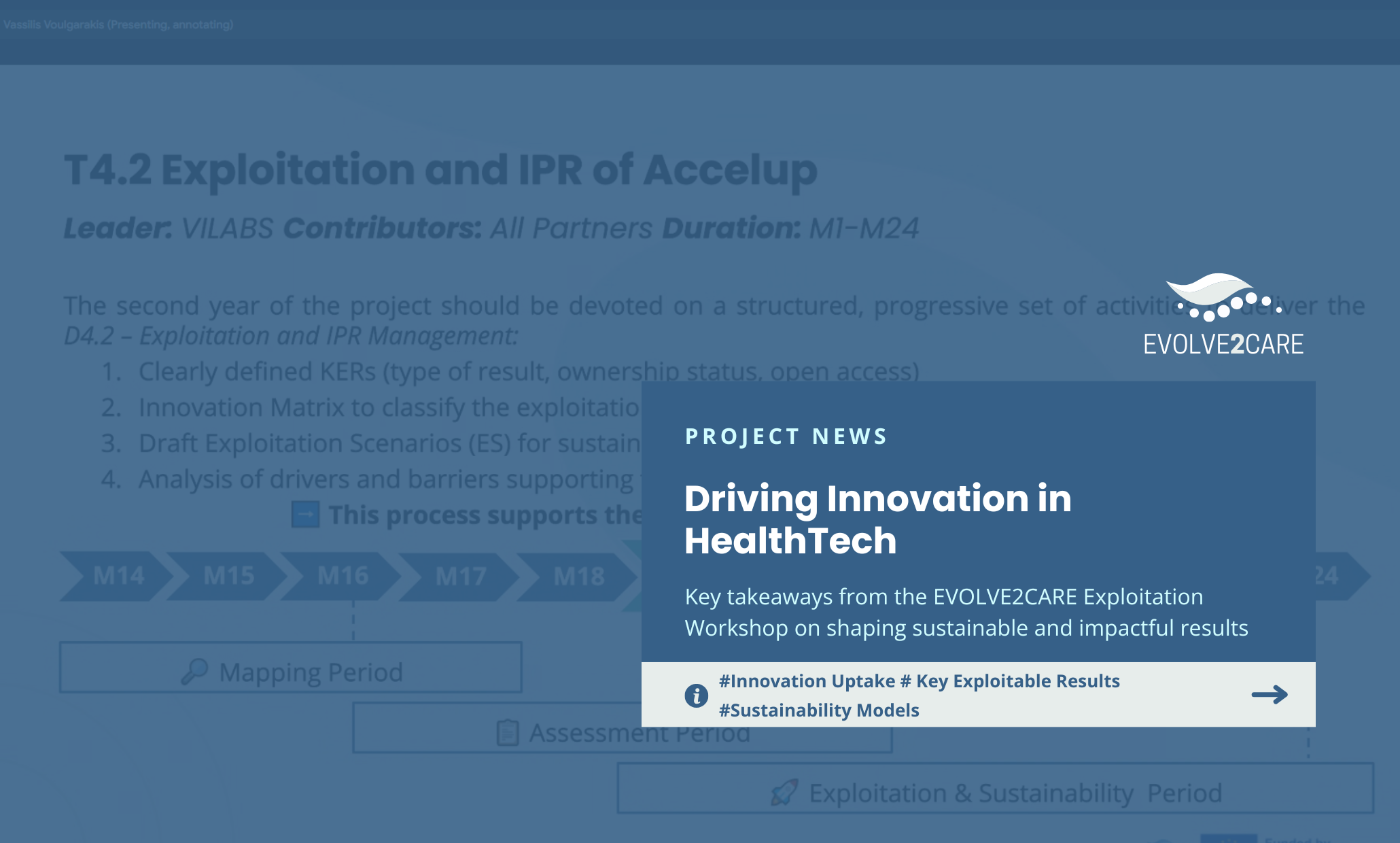
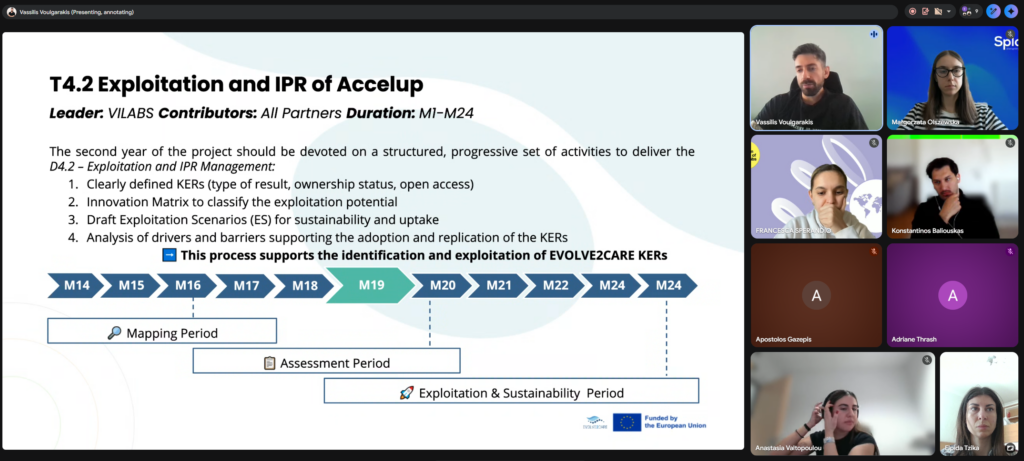
The journey from a research project to real-world impact requires a clear map. Recently, the EVOLVE2CARE team gathered for an intensive internal workshop to define that very map. The goal was simple but ambitious: to identify the project’s Key Exploitable Results (KERs) and establish a robust sustainability strategy to ensure our innovations thrive long after the project concludes.
This session marked a pivotal shift from development to delivery, focusing on how our outputs, ranging from digital platforms to policy frameworks, will serve the Health Tech Ecosystem in the years to come.
Mapping the Path: Four Key Exploitable Results
Through a participatory analysis, the team identified four core KERs. These elements are not just standalone products. The EVOLVE2CARE KERs form a complementary pathway from innovation to scaling.
- AccelUP Market Platform: Our most technical asset, this digital platform serves as the central hub connecting innovators with Living Labs. It is the primary engine for commercialization and long-term sustainability.
- KPI Framework for Health Tech Evaluation: A structured methodology designed to measure the real-world impact of health tech solutions, providing the evidence-based validation that investors and healthcare providers demand.
- Training and Capacity Building Program: A training program that equips Living Labs with essential skills in service design and experimentation, currently hosted via the ENoLL Academy.
- Living Lab Experimentation and Validation Framework: A unified methodology for designing and implementing health tech experiments, ensuring scalability and replication across different healthcare environments.
Strategizing for Sustainability: Commercial vs. Open Access
A major focus of the workshop was classifying these results into Tiers to determine their commercialisation nature. We categorized them based on their primary value:
Tier 1: Commercial Excellence
The AccelUP Platform remains the core commercial asset. Future efforts will focus on dedicated workshops to refine its business model, ensuring it becomes a self-sustaining marketplace for health tech stakeholders.
Tier 2: Non-Commercial & Cross-Cutting Impact
For results like the KPI Framework and the Training Program, the team reached a consensus: Open Access is key. Additionally, it was agreed that the Training Program will remain free of charge to maximize reach among researchers, innovators, and policymakers.
Building for the Future
Sustainability is only possible if the work remains relevant. During the session, partners discussed the importance of keeping training materials “living” by exploring ways to expand content rather than just maintaining it. We also committed to a dissemination strategy that moves beyond traditional repositories like Zenodo to engage directly with scientific communities and European decision-makers.
What’s Next?
The work does not stop here. The team is currently refining the exploitation strategy and formalising the sustainability models, with the aim of presenting the Key Exploitable Results of EVOLVE2CARE in the near future.
By standardizing how we measure impact and build capacity, EVOLVE2CARE is setting a new benchmark for how Health Tech Innovation can be validated and scaled across Europe.
Stay tuned as we move into the final stages of the project, bringing these tools to the wider health tech community!